January 4, 2024
Research has shown that both optometrists and ophthalmologists frequently fail to diagnose age-related macular degeneration (AMD) about 25% of the time with no difference between the two specialties.1 Histopathological and clinical research suggests that subclinical AMD has both structural and functional consequences before drusen are even evident in clinical imaging.2-5 In fact, drusen represent the tip of the iceberg of the earliest changes caused by AMD. What if practitioners could not only identify those with early AMD by dark adaptation testing before drusen develop but also better identify those most at risk of developing the disease before it begins by macular pigment optical density (MPOD) testing and careful case histories such as family history and lifestyle factors? Educating patients on modifiable lifestyle factors at an earlier stage before drusen appear could change the trajectory of the disease and save sight.
In an article for Women in Optometry, Professor of Optometry Dr. Julie Rodman makes a clear case for earlier identification of macular degeneration through dark adaptation testing. In her article, “Big Change Is a Small Choice,” Professor Rodman points out that trouble seeing at night is a symptom that appears before the onset of drusen, and impaired dark adaptation is an early symptom. Waiting until drusen appears is a broken system as so many people continue to go blind from the disease. “It’s broken, so let’s fix it…by detecting AMD before clinical signs are evident on examination and managing early and intermediate stage disease with the same close attention that retina specialists focus on late-stage disease, we potentially can make a significant difference in patients’ lives.”
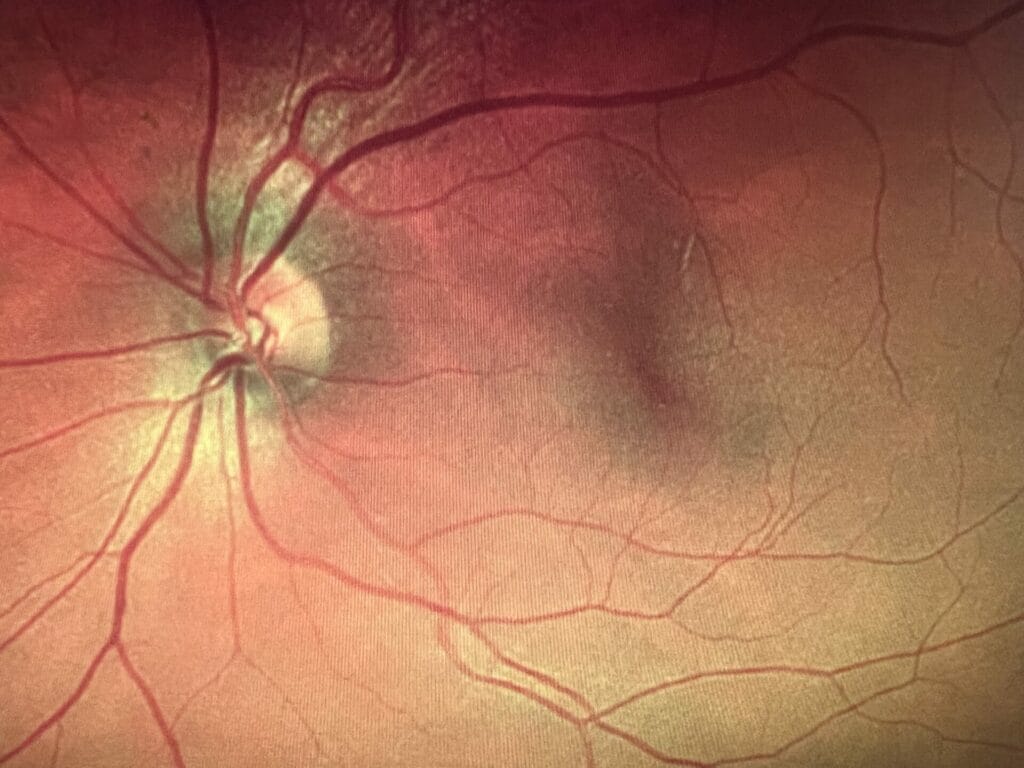
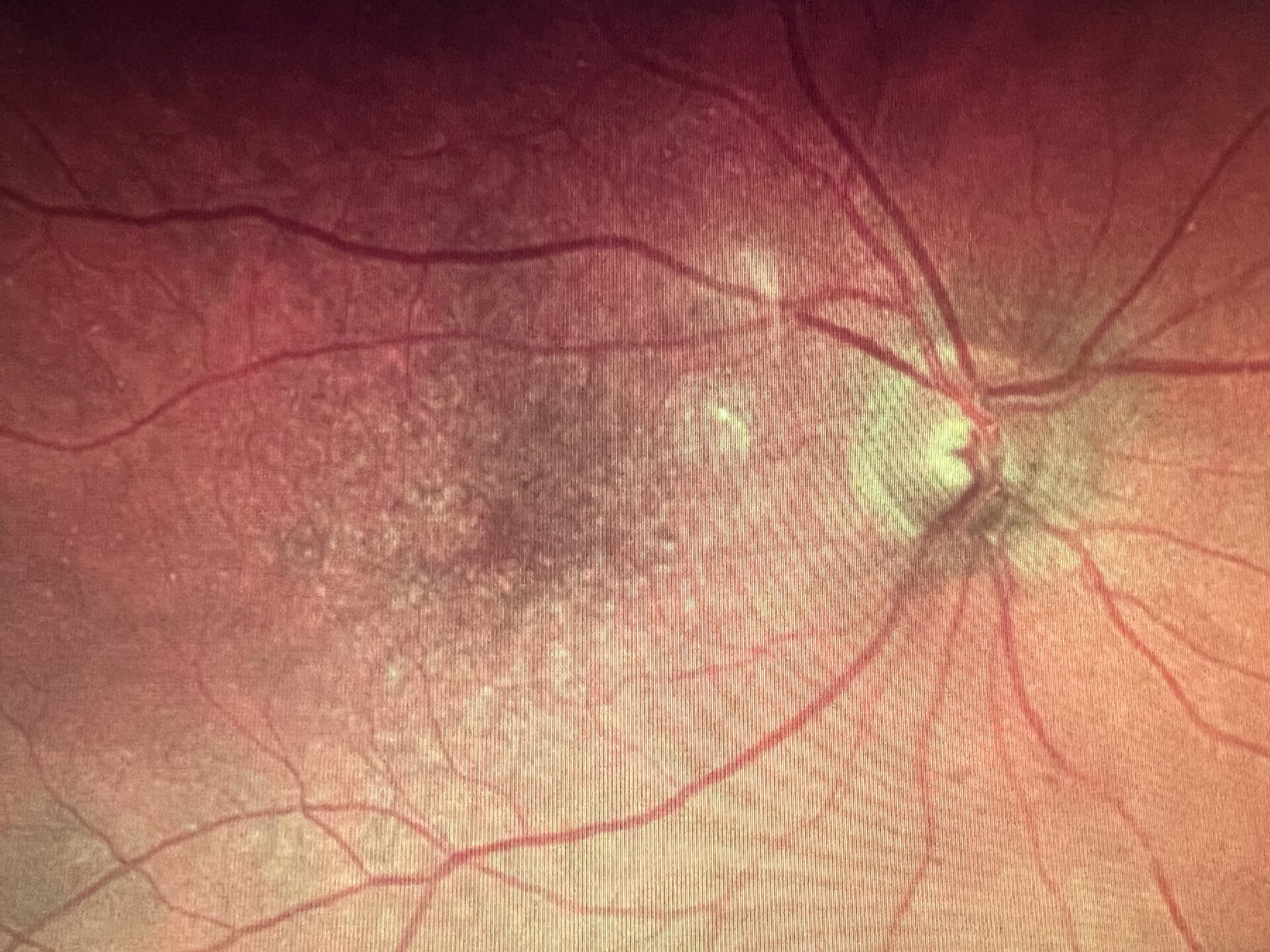
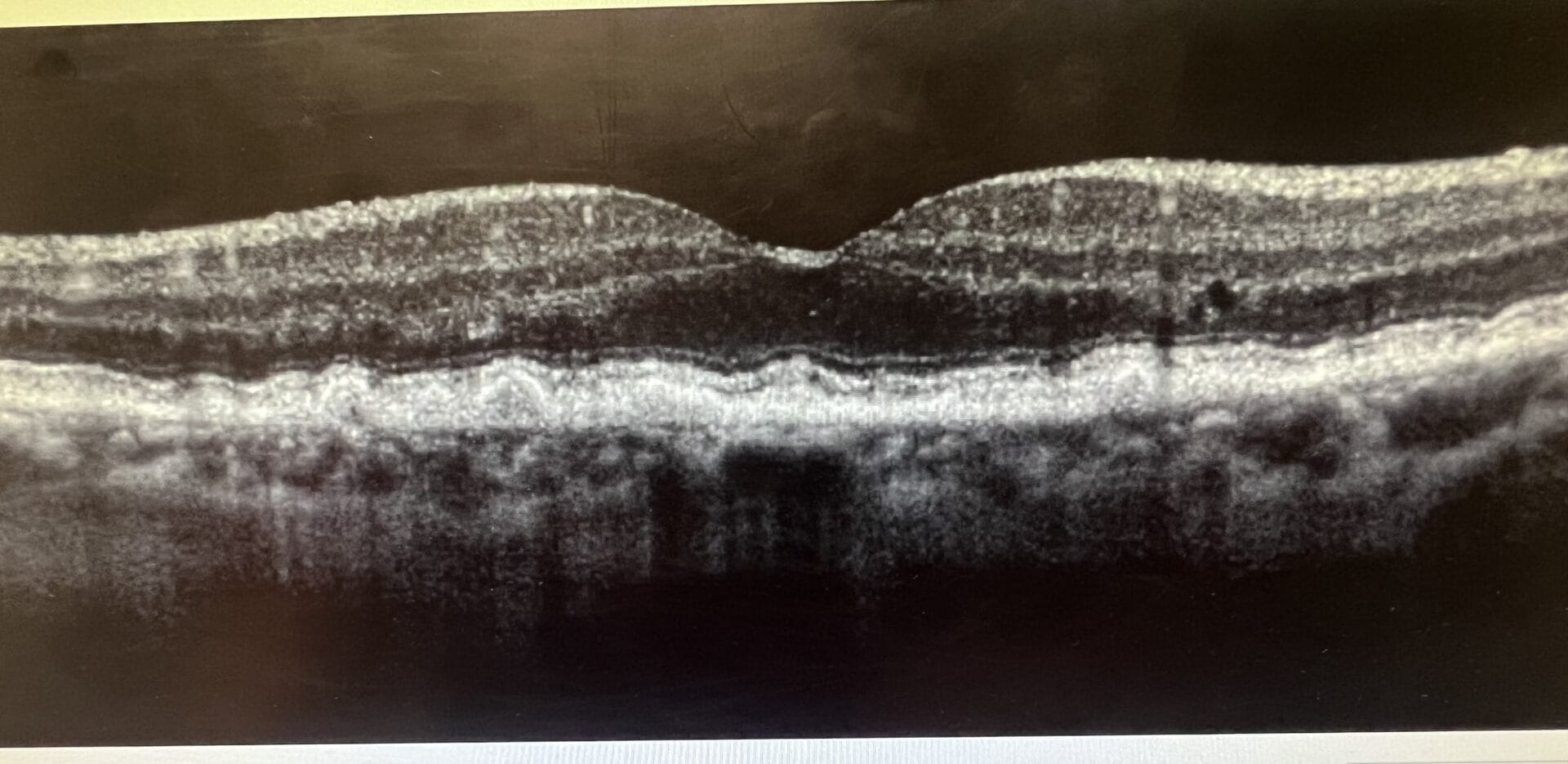
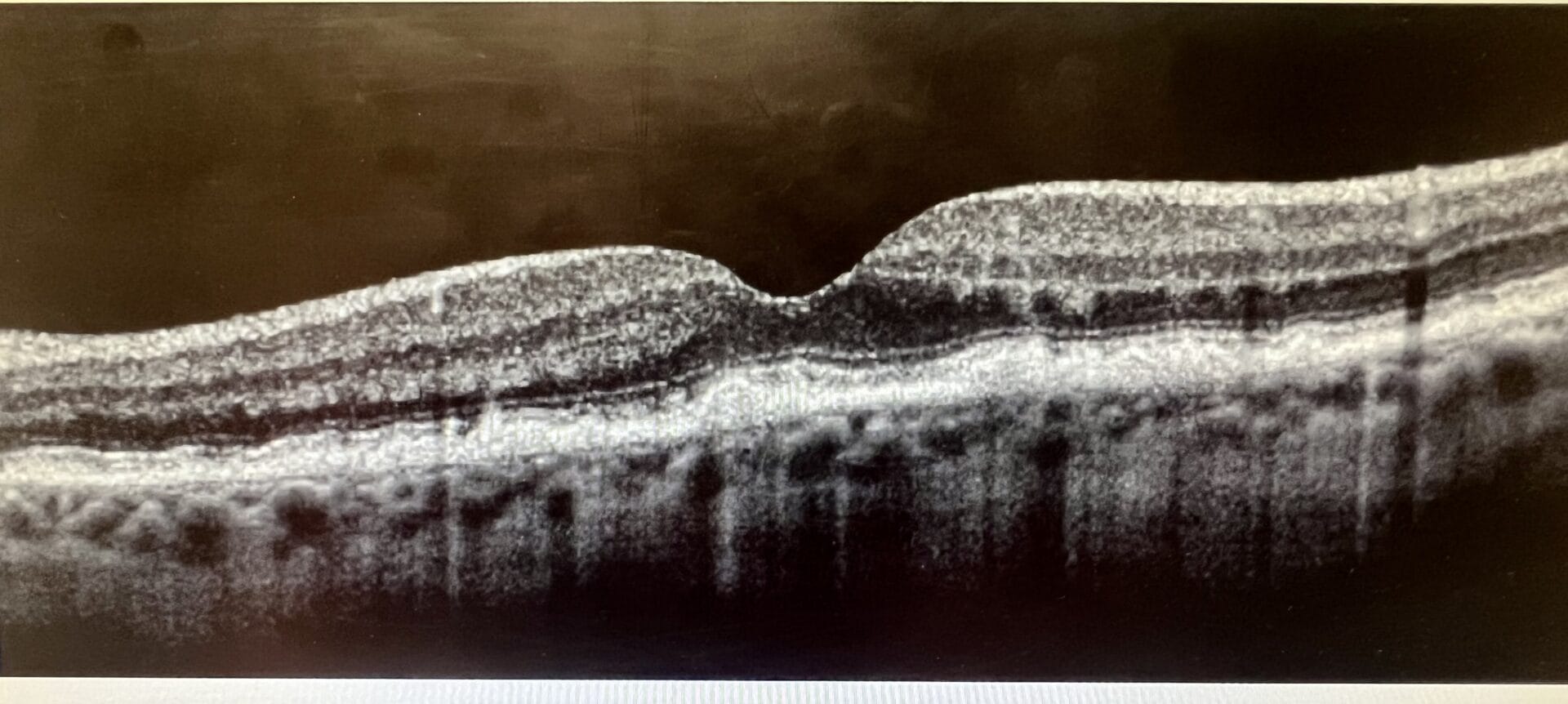
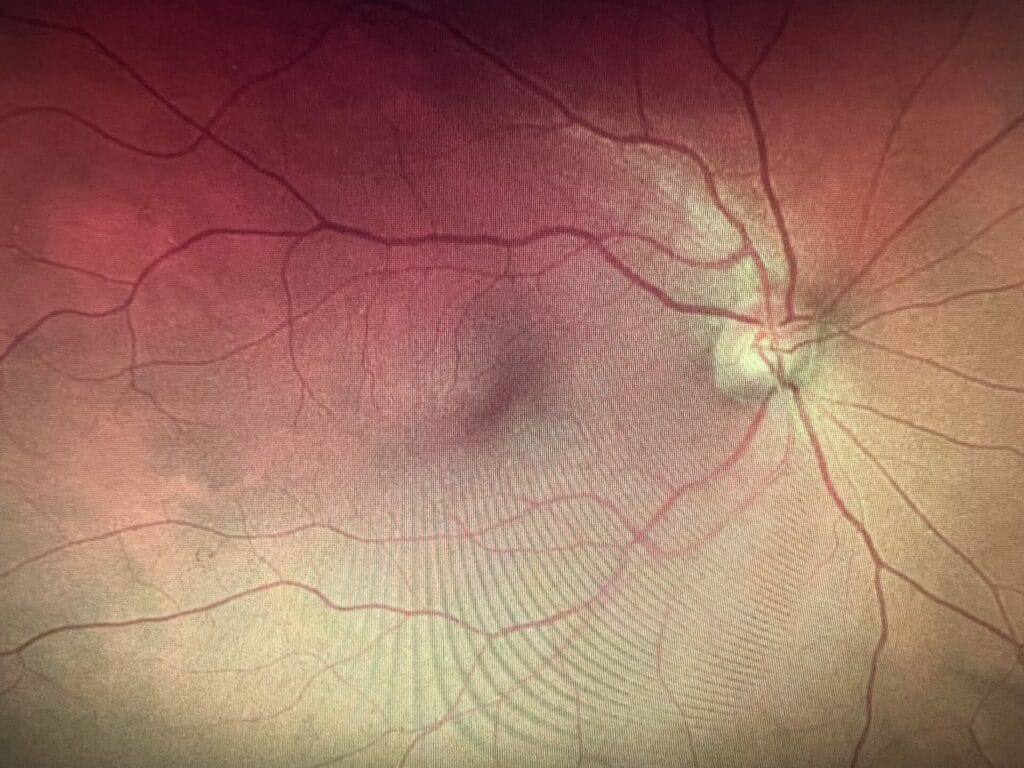
Optomap and OCT of both eyes from a 78-year-old female with intermediate to advanced dry AMD. This patient is on an AREDS2 supplement with fatty fish consumption twice a week and a Mediterranean diet. She has been advised of evidence-based lifestyle interventions to help reduce the risk of disease progression.
Discuss Prevention Strategies
Eye doctors can learn a lot about prevention from the American Heart Association. When heart experts talk about prevention, they usually refer to one of three types: secondary, primary, and primordial prevention.6,7
Secondary prevention: These efforts are started after someone has already had a heart attack or stroke, undergoes bypass surgery, or develops some other form of heart disease in an attempt to prevent a second heart attack or stroke.
Primary prevention: This type of intervention aims to keep an individual at risk of heart disease due to risk factors such as high blood pressure or high cholesterol from having a first heart attack or stroke. The appearance though of worrisome cardiovascular risk factors means that inflammation, atherosclerosis, and/or endothelial dysfunction are already at work.6
Primordial prevention: The word “primordial” means existing from the beginning, and this type of prevention involves working to prevent inflammation, atherosclerosis, and endothelial dysfunction from taking hold in the first place. Once rarely discussed, primordial prevention is now the cornerstone of the American Heart Association’s definition of ideal heart health and efforts to help people achieve it.6 As its name applies, the sooner you can start practicing primordial prevention, ideally from childhood on, the likely you are to achieve it and protect yourself from heart disease.6, 7
If we take these prevention terms and apply them to irreversible vision loss from the most common cause of blindness in individuals over 50 in the developed world, advanced macular degeneration, it may look something such as the following:
Secondary prevention would apply to the sight-saving injections for late-stage AMD that retinal surgeons use to stave off further vision loss once the advanced stage has occurred.
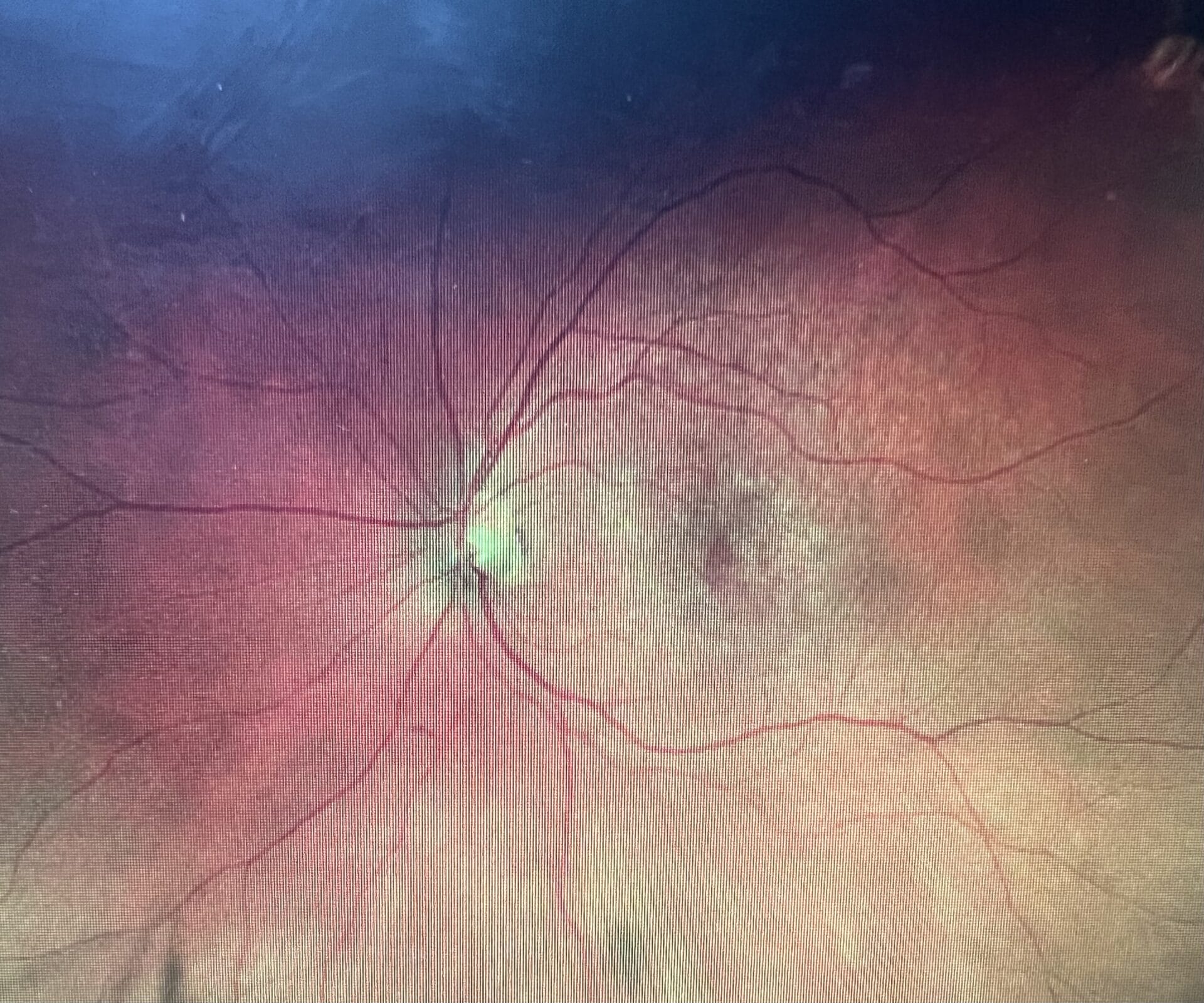
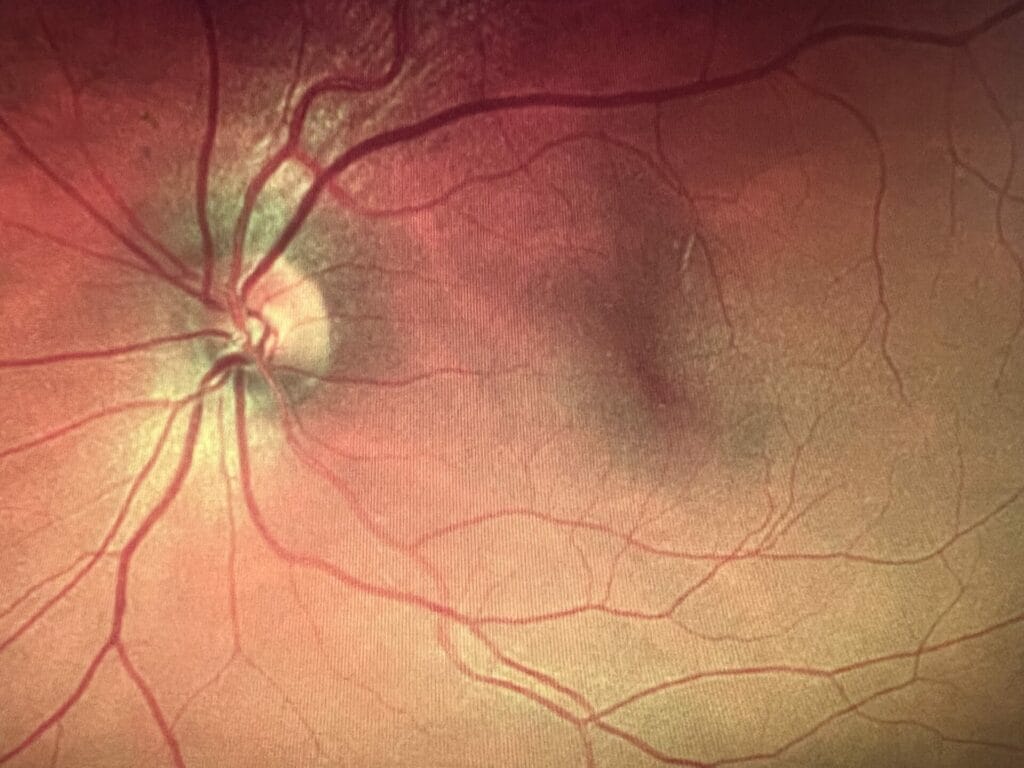
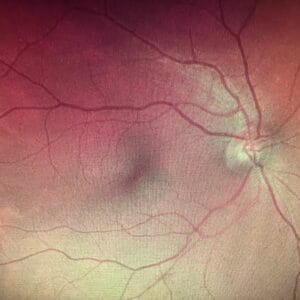
Optomap from 78-year-old patient’s 57-year-old son with no macular drusen and no night driving complaints. Because of his family history of macular degeneration, in-office MPOD testing was performed, and his score was 0.20, which is very low and falls into the category of grossly inadequate photoreceptor protection. This patient was put on a macular carotenoid supplement and advised on lifestyle prevention measures to reduce his risk of AMD.
Primary prevention could apply to the use of AREDS2 supplements for intermediate or late AMD where the formation of drusen has already occurred. Research shows that RPE cells deposit locally generated cholesterol beneath the RPE cell layer and in Bruch’s membrane before drusen are formed.2,5 With disease progression, this cholesterol continues to accumulate, resulting in focal areas that are thickened enough to be identified as drusen.2,3,4,5 This cholesterol accumulation causes three primary insults to the retina: inflammation, oxidative stress, and disruption of oxygen and nutrition supplies to the outer retina.3,5 Waiting until intermediate AMD to intervene with AREDS2 supplements means that inflammation and oxidative stress have been occurring for some time.
In his opinion as stated in an article he wrote on how early diagnosis can improve AMD outcomes, Dr. Jeffry Gerson, who lectures and writes extensively on AMD, stated the following, “The clinical staging of AMD is misinterpreted and has led to a defeatist view of what we can achieve in terms of more effective management of dry AMD. By the time we can visualize change, damage has already been done.” 5
Primordial prevention could include public health interventions with the aim to adopt a correct lifestyle, including healthy diet, physical activity, and cessation of smoking as recommended strategies for AMD prevention. Educating patients with a family history of macular degeneration on prevention strategies should occur with every eye examination.
Nutrients and Food Sources for AMD Prevention9
| Nutrients | Food Source |
| Vitamin C | Citrus fruits and juices, green peppers, broccoli, potatoes |
| Vitamin E | Whole grains, eggs, nuts, collard greens, spinach |
| Beta carotene | Carrots, kale, spinach |
| Zinc | Meat, poultry, fish, whole grains, oysters, nuts, eggs |
| Lutein and zeaxanthin | Spinach, kale, egg yolk, parsley, other green leafy vegetables |
| Omega-3 (DHA and EPA) | Fish and seafood, nuts and seeds, fortified foods |
Are Leafy Greens the New Carrots of Healthy Vision?
Carrots have long been considered the go-to vegetable for healthy vision. Rich in beta carotene that is converted to vitamin A and needed to form rhodopsin, carrots provide vital nutrients for the retina. However, the association between carrots and eyesight originated from a myth. During World War II, British Royal Air Force pilots began using radar to target and shoot down enemy planes. In an effort to keep this new technology a secret, the visual accuracy of the pilots, especially at night, was attributed to eating carrots. This led to a longstanding campaign that promoted carrots for better eyesight. Based on emerging research on the benefits of the dietary carotenoids lutein, zeaxanthin, and meso-zeaxanthin, a more accurate public health campaign today would promote leafy greens for healthy vision.
Studies such as The Blue Mountains Eye Study show a greatly reduced risk of developing drusen or AMD with higher amounts of lutein and zeaxanthin in the diet.10,11,12 A study by Huang et al. showed that participants with high levels of serum lutein and zeaxanthin had a 79% reduced risk of developing AMD in comparison to subjects with lower serum concentration.13 It has been shown that in the aging retina, the inhibition of the endogenous antioxidant capacity, marked by the depletion of macular carotenoids lutein, zeaxanthin, and meso-zeaxanthin, is a major contributing factor in the progression of AMD.14 Thus, discussing the addition of more leafy greens in the diet for better eye health is justified on every patient.
Interestingly, higher macular pigment scores are not only applicable to the prevention of macular degeneration. In a recent study from the National Institutes of Health, researchers showed that macular pigment density may indicate a new risk factor for glaucoma.15 The researchers observed a positive association between MPOD and central subfield GCC thickness 15 years later. MPOD was positively associated with peripapillary RNFL superior and temporal quadrant thickness among eyes with primary open angle glaucoma. Their results linked low MPOD to retinal layers that are structural indicators of early glaucoma.15
Sum of the Parts: Lifestyle Factors Beyond Leafy Greens
A single nutrient or food approach is not able to reach the synergistic effects of food and nutrients consumed in combination in the diet, therefore overall dietary patterns have been studied. A reduced risk of all types of AMD has been shown with a Mediterranean Diet.16,17 Along with diet, other factors such as smoking cessation, regular physical activity, reducing excessive body weight and avoiding obesity, and sleeping seven to eight hours per night have all been associated with a decreased risk of developing AMD.8 Concerning body mass index (BMI), it is important to stress that studies have shown that excess body fat can alter the transport and deposition of carotenoids from blood to the macula, leading to a reduction of macular pigment levels in the fovea.8,18 Excess body weight and obesity are associated with a higher risk of developing AMD, and it has been demonstrated that people affected by obesity have a major risk to manifest late AMD in comparison to subjects with a normal weight.8,19
We as a profession can no longer ignore the science of prevention for not only macular degeneration but also other blinding eye diseases such as glaucoma. Minding the macular pigment through addressing the importance of green leafy vegetables in our diets as well as overall eating patterns has been shown to be preventative for one of the most blinding eye diseases on the planet. Public health campaigns successfully create awareness and help evoke change. Our current health care system of waiting until disease is apparent before intervening is broken and not sustainable. Because of our imaging technologies, we as a profession are perfectly poised to be on the forefront of prevention. Let’s hope in 2024 there exists a greater awareness amongst doctors and patients alike of the power of nutrition and lifestyle to mitigate the risk and course of disease.
References
1 Owsley C, McGwin G, Clark M, et al. Delayed rod-mediated dark adaptation is a functional biomarker for incident early age-related macular degeneration. Ophthalmology, October 30, 2015.
2 Pikuleva I, Curcio C. Cholesterol in the retina: the best is yet to come. Progress in Retinal and Eye Research. 2014 July:41;64-89.
3 Owsley C, McGwin G, Jackson G, et al. Effect of short-term, high-dose retinol on dark adaptation in aging and early age-related maculopathy. Invest Ophthalmol Vis Sci. 2006:47(4);1310-8.
4 Owsley C, Jackson G, White M, et al. Delays in rod-mediated dark adaptation in early age-related maculopathy. Ophthalmology. 2001;108(7):1196-202.
5 Gerson J. How early diagnosis can improve AMD outcomes. Review of Optometry. September 15, 2017.
6 Lloyd-Jones DM, Hong Y, Labarthe D, et al. Defining and setting national goals for cardiovascular health promotion and disease reduction: the American Heart Association’s strategic Impact Goal through 2020 and beyond. Circulation, 2010;121:586-613.
7 Harvard T.H. Chan School of Public Health. The Nutrition Source. Preventing Heart Disease.harvard.edu
8 DiCarlo E, Augustin A. Prevention of the onset of age-related macular degeneration. Clin. Med. 2021, 10, 3297.
9 Van Leeuwen R, Boekhoorn S, Vingerling JR, et al. Dietary intake of antioxidants and risk of age-related macular degeneration. JAMA 2005, 294, 3101-3107.
10 Tan JSL, Wang JJ, Flood V, et al. Dietary fatty acids and the 10-year incidence of age-related macular degeneration: The Blue Mountains Eye Study. Arch Ophthalmol. 2009, 127, 656-665.
11 Walchuk C, Suh M. Nutrition and the aging retina: A comprehensive review of the relationship between nutrients and their role in age-related macular degeneration and retina disease prevention. Food Nutr. Res. 2020, 93,293-332.
12 Delcourt C, Carriere L, Delage M, et al. POLA Study Group Plasma lutein and zeaxanthin and other carotenoids as modifiable risk factors for age-related maculopathy and cataract: The POLA Study. Ophthmaol. Vis. Sci. 2006, 47, 2329-2335.
13 Huang LL, Coleman HR, Kim J et al. Oral supplementation of lutein/zeaxanthin and omega-3 long chain polyunsaturated fatty acids in persons aged 60 years and older, with or without AMD. Ophthmaol. Vis. Sci.2008, 49, 3864-3869.
14 Lem DW, Gierhart DL, Davey PG. A systematic review of carotenoids in the management of diabetic retinopathy. 2021 Jul; 13(7):2441.
15 Lawler T, Mares JA, Liu Z et al. Carotenoids in age-related eye disease study investigators. “Association of macular pigment optical density with retinal layer thickness in eyes with and without manifest primary open-angle glaucoma.” October 6, 2023 BMJ Open Ophthalmology.
16 Merle BM, Colijn JM, Cougnard-Gregoire A, et al. EYE-RISK Consortium. Mediterranean Diet and Incidence of Advanced Age-Related Macular Degeneration: The EYE-RISK Consortium. Ophthalmology. 2019;126:381-390.
17 Nunes S, Alves D, Barreto P et al. Adherence to a Mediterranean diet and its association with age-related macular degeneration. The Coimbra Eye Study report 4. Nutrition 2018:51:6-12.
18 Bovier ER, Lewis RD, Hammond BR. The relationship between lutein and zeaxanthin status and body fat. Nutrients, 2013;5:750-757.
19 Zhang QY, Tie LJ, Wu SS et al. Overweight, obesity, and risk of age-related macular degeneration. Ophthalmol. Vis. Sci. 2016;57:1276-1283.